浏览量: 764
- 产品名称: Navoximod
- 产品货号: CS0071
- 货期: 现货
- 价格与订购: 1566
- 数量:
库存: 100
- 规格: 1mg 5mg
- 产品信息
- 如何订购
产品描述
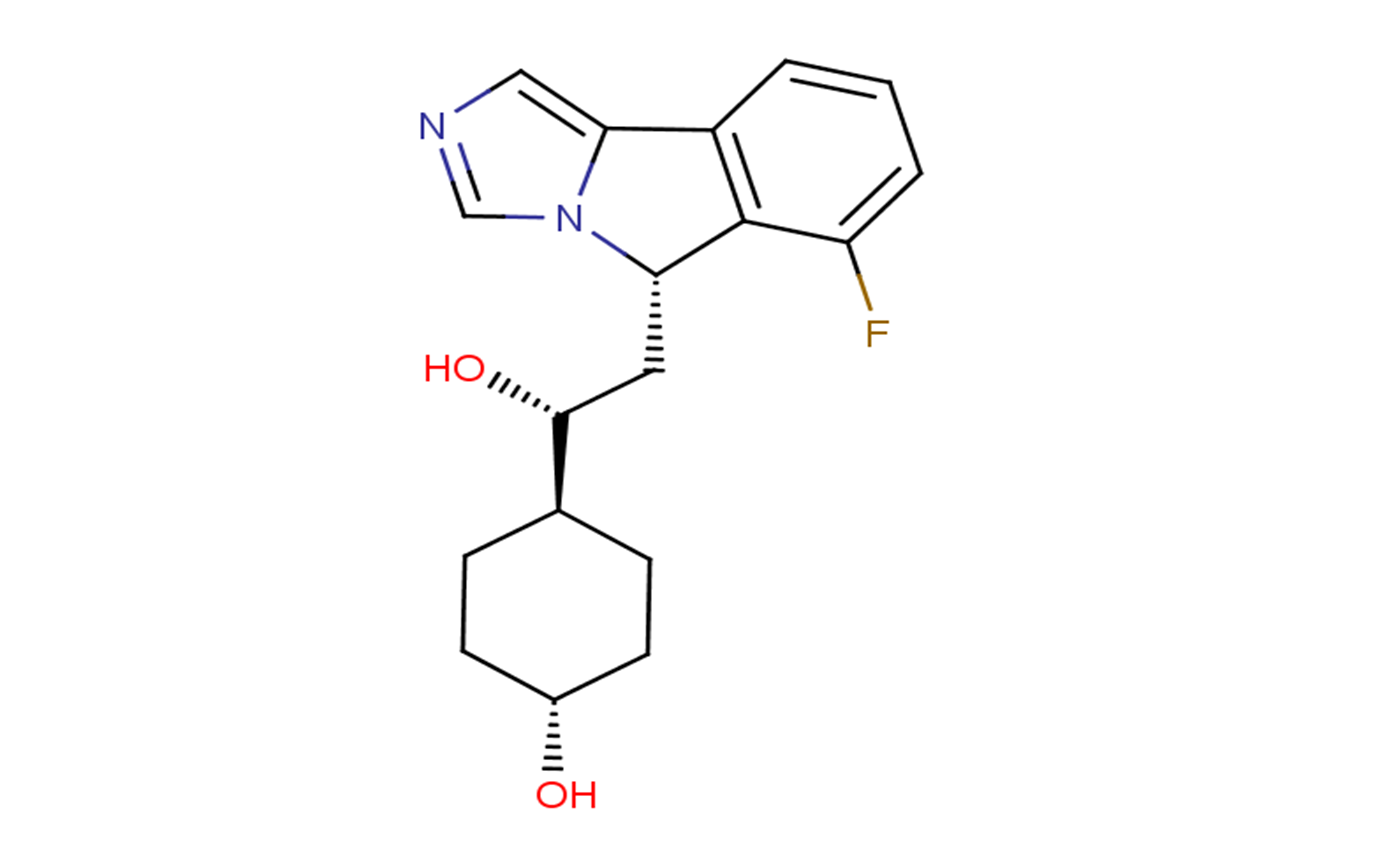
Navoximod (NLG-919, GDC-0919) is a potent indoleamine-(2,3)-dioxygenase (IDO) pathway inhibitor (Ki/EC50: 7 nM/75 nM).
体外活性
Using IDO-expressing human monocyte-derived dendritic cells (DCs) in allogeneic mixed lymphocyte reaction (MLR) reactions, Navoximod potently blocks IDO-induced T cell suppression and restores robust T cell responses (ED50: 80 nM). Similarly, using IDO-expressing mouse DCs from tumor-draining lymph nodes, Navoximod abrogates IDO-induced suppression of antigen-specific T cells (OT-I) in vitro (ED50: 120 nM) [1]. Navoximod inhibits the IDO activity in a concentration-dependent manner with an EC50 of 0.95 μM. PEG2k-Fmoc-NLG(L) is less active (EC50: 3.4 μM) in inhibiting IDO compared with free Navoximod while PEG2k-Fmoc-NLG(S) is least active (EC50>10 μM). Coculture of IDO+tumor cells with splenocytes isolated from BALB/c mice leads to significant inhibition of T-cell proliferation. This inhibition is significantly attenuated when the mixed cells are treated with Navoximod. PEG2k-Fmoc-NLG(L) is also active in reversing the inhibitory effect of tumor cells although slightly less potent than Navoximod [3].
体内活性
In mice, a single oral administration of Navoximod reduces the concentration of plasma and tissue Kyn by ~50%. In vivo, in mice bearing large established B16F10 tumors, administration of Navoximod markedly enhances the anti-tumor responses of naïve, resting pmel-1 cells to vaccination with cognate hgp100 peptide plus CpG-1826 in IFA. In this stringent established-tumor model, Navoximod plus pmel-1/vaccine produces a dramatic collapse of tumor size within 4 days of vaccination (~95% reduction in tumor volume compared to control animals receiving pmel-1/vaccine alone without Navoximod) [1]. When combined with Temozolomide (TMZ)+radiation therapy (RT), both Navoximod and 1-methyl-D-tryptophan (D-1MT, indoximod) enhance survival relative to mice treated with TMZ+RT alone [2].
细胞实验
Briefly, HeLa cells are seeded in a 96-well plate at a cell density of 5000 cells per well and allowed to grow overnight. Recombinant human IFN-γ is then added to each well with a final concentration of 50 ng/mL. At the same time, various concentrations of PEG2k-Fmoc-NLG(L), PEG2k-Fmoc-NLG(S) or Navoximod (NLG919) (50 nM-20 μM) are added to the cells. After 48 h of incubation, 150 μL of the supernatants per well is transferred to a new 96-well plate. Seventy-five μL of 30% trichloroacetic acid is added into each well and the mixture is incubated at 50°C for 30 min to hydrolyse N-formylkynurenine to kynurenine. For the colorimetric assay, supernatants are transferred to a new 96-well plate, mixed with an equal volume of Ehrlich reagent (2% p-dimethylamino-benzaldehyde w/v in glacial acetic acid), and incubated for 10 min at RT. The reaction product is measured at 490 nm by a plate reader [3].
动物实验
Mice are anesthetized with 4% isoflurane, and the surgical plane of anesthesia is maintained with 2% isoflurane in oxygen. Mice are immobilized in a stereotactic frame for tumor implantation. Briefly, the skull is shaved and exposed with a 0.5 cm skin incision. With the antiseptic technique, 10^5 GL261 cells (suspended in 3 μL RPMI-1640) are injected at the following coordinates with respect to the bregma on the right side (antero-posterior, -2 mm; medio-lateral, 2 mm; dorsoventral, 3 mm). This placement reproducibly yielded tumor growth in a paracortical area of the posterolateral right frontal lobe. Tumor-bearing mice are treated with combinations of oral DL-1MT (2 mg/mL D-1MT mixed with 2 mg/mL L-1MT) in drinking water, D-1MT (4 mg/mL) in drinking water, Navoximod (6 mg/mL) in drinking water, intraperitoneal cyclophosphamide, intraperitoneal temozolomide, and/or total-body radiation (500 cGy from a 137Cs source), as detailed in figure legends. Mice are observed daily and sacrificed when they became ill or moribund [2].
参考文献
1. Mario R. Mautino, et al. Abstract 491: NLG919, a novel indoleamine-2,3-dioxygenase (IDO)-pathway inhibitor drug candidate for cancer therapy. AACR 104th Annual Meeting 2013; Apr 6-10, 2013.
2. Li M, et al. The indoleamine 2,3-dioxygenase pathway controls complement-dependent enhancement of chemo-radiation therapy against murine glioblastoma. J Immunother Cancer. 2014 Jul 7;2:21.
3. Chen Y, et al. An immunostimulatory dual-functional nanocarrier that improves cancer immunochemotherapy. Nat Commun. 2016 Nov 7;7:13443.
别名
GDC-0919
纯度
99.81%
分子量
316.37
纯度
>95% as determined by SDS-PAGE
分子式
C18H21FN2O2
CAS No
1402837-78-8
存储
0-4℃ for short term (days to weeks), or -20℃ for long term (months).
溶解度
DMSO: 100 mg/mL (316.09 mM), Need ultrasonic
( < 1 mg/ml refers to the product slightly soluble or insoluble )
icon Note
For research use only .
上一篇 Pexidartinib

 地 址:
地 址: 产品销售:
产品销售: E - mail :
E - mail : 邮 编:
邮 编:
 Amily
Amily


